
| Size | Price | Stock | Qty |
|---|---|---|---|
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| Other Sizes |
Purity: ≥98%
Almorexant HCl (also known as ACT-078573; ACT078573), the hydrochloride salt of Almorexant, is a novel, potent, orally bioactive, competitive and dual orexin1/2 receptor antagonist indicated for the treatment of sleep disorders/insomnia. It inhibits OX1 and OX2 receptors with IC50s of 6.6 nM and 3.4 nM, respectively. Almorexant exhibits competitive antagonistic effects on hOX1R and noncompetitive-like effects on hOX2R in the inositol phosphates assay. Almorexant also has an impact on sleep in a variety of species, including humans. In normal C57BL/6 mice, it dose-dependently shortens the amount of time spent awake and lengthens the amount of time spent in NREM and REM sleep.
| Targets |
human OX2R ( Kd = 0.17 nM ); human OX1R ( Kd = 1.3 nM ); Caspase-3
Almorexant HCl (ACT 078573) targets orexin 1 receptor (OX1R) and orexin 2 receptor (OX2R), with a Ki value of 0.5 nM for human recombinant OX1R and 0.3 nM for human recombinant OX2R [2] Almorexant HCl (ACT 078573) has an IC50 value of 1.2 nM for rat OX1R and 0.8 nM for rat OX2R [2] |
|---|---|
| ln Vitro |
In vitro activity: Almorexant inhibits the rise in intracellular Ca2+ that is brought about by 10 nM human orexin-A in Chinese hamster ovarian cells, with IC50 values for the OX1 and OX2 receptors of 16 nM (rat) and 13 nM (human), respectively.[1]
Almorexant HCl (ACT 078573) concentration-dependently inhibits orexin-A-induced proliferation of human pancreatic cancer cells (PANC-1, MIA PaCa-2), achieving a 52% proliferation inhibition rate in PANC-1 cells and 48% in MIA PaCa-2 cells at 10 μM [1] Almorexant HCl (ACT 078573) promotes apoptosis of pancreatic cancer cells; after 48 hours of treatment at 10 μM, the apoptosis rate of PANC-1 cells increases from 3.2% (control group) to 18.5%, accompanied by downregulation of Bcl-2 protein expression and upregulation of Bax protein expression [1] Almorexant HCl (ACT 078573) blocks orexin-A-induced calcium influx in rat hypothalamic neurons, with an inhibition rate of 70% at 1 nM and complete inhibition at 5 nM [2] Almorexant HCl (ACT 078573) significantly inhibits OX2R-mediated signaling pathway induced by orexin-B, with an IC50 value of 0.6 nM [2] |
| ln Vivo |
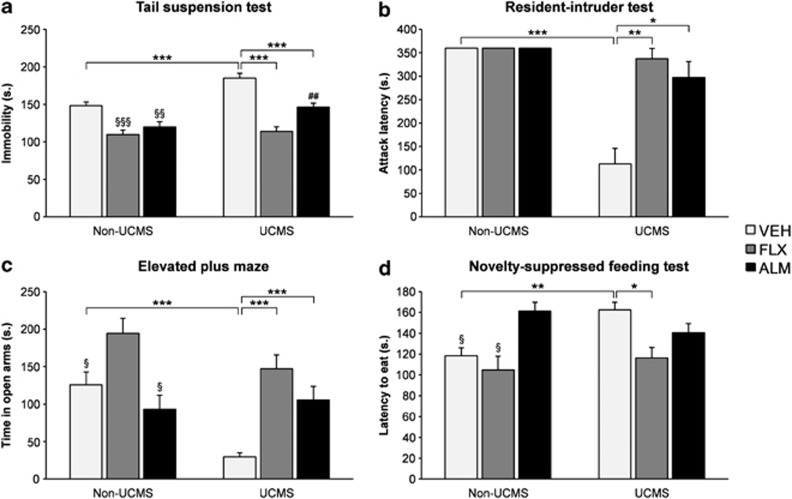
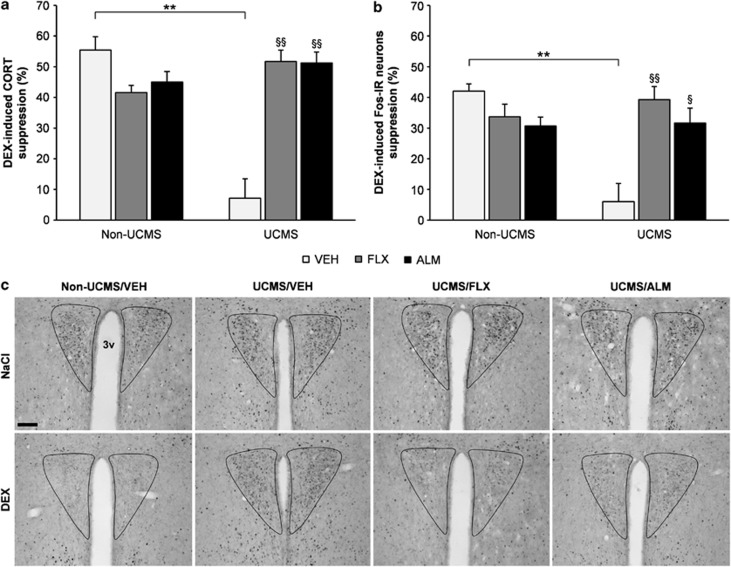
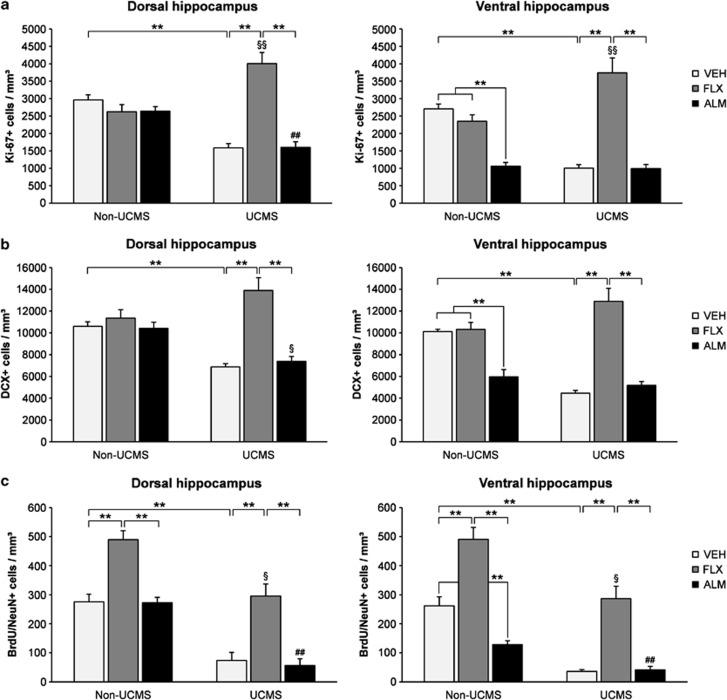
Almorexant (300 mg/kg p.o.) increases electrophysiological indices of both REM and non-REM sleep and decreases alertness in male Wistar rats. Almorexant (100 mg/kg p.o.) increases surrogate markers of REM sleep and induces somnolence in dogs.[1] Without causing neurogenesis, almorexant produces a strong antidepressant-like effect and restores the HPA axis defect caused by stress.[2] Furthermore, in models of high-drinking rodents, Almorexant also decreases ethanol self-administration.[3]
Almorexant HCl (ACT 078573) orally administered to nude mice bearing PANC-1 pancreatic cancer xenografts (50 mg/kg/day for 21 consecutive days) reduces tumor volume by 45% and tumor weight by 42% compared with the control group, without significant weight loss [1] Almorexant HCl (ACT 078573) intraperitoneally injected at 10 mg/kg into orexin knockout (orexin-/-) narcoleptic mice prolongs total sleep time by 30% and increases cataplexy attack frequency by 2.3-fold [3] Almorexant HCl (ACT 078573) orally administered to SD rats at 3, 10, 30 mg/kg/day for 14 consecutive days shows no significant differences in escape latency or target quadrant residence time compared with the control group in the Morris water maze test, indicating no impairment of learning and memory function [4] Almorexant HCl (ACT 078573) orally administered to C57BL/6 mice at 20 mg/kg significantly shortens sleep latency (from 18.2 minutes to 8.5 minutes), prolongs non-rapid eye movement (NREM) sleep time, and has no obvious effect on rapid eye movement (REM) sleep [5] Almorexant HCl (ACT 078573) orally administered to rhesus monkeys at 10 mg/kg reduces nighttime wake time by 40% and increases sleep efficiency from 72% to 88% [6] |
| Enzyme Assay |
Recent preclinical and clinical research has shown that Almorexant promotes sleep in animals and humans without disrupting the sleep architecture. Here, the pharmacology and kinetics of [(3)H]Almorexant binding to human orexin 1 receptor (OX(1))- and human orexin 2 receptor (OX(2))-human embryonic kidney 293 membranes were characterized and compared with those of selective OX(1) and OX(2) antagonists, including 1-(5-(2-fluoro-phenyl)-2-methyl-thiazol-4-yl)-1-((S)-2-(5-phenyl-(1,3,4)oxadiazol-2-ylmethyl)-pyrrolidin-1-yl)-methanone (SB-674042), 1-(6,8-difluoro-2-methyl-quinolin-4-yl)-3-(4-dimethylamino-phenyl)-urea (SB-408124), and N-ethyl-2-[(6-methoxy-pyridin-3-yl)-(toluene-2-sulfonyl)-amino]-N-pyridin-3-ylmethyl-acetamide (EMPA). The effect of these antagonists was also examined in vitro on the spontaneous activity of rat ventral tegmental area (VTA) dopaminergic neurons. [(3)H]Almorexant bound to a single saturable site on hOX(1) and hOX(2) with high affinity (K(d) of 1.3 and 0.17 nM, respectively). In Schild analyses using the [(3)H]inositol phosphates assay, Almorexant acted as a competitive antagonist at hOX(1) and as a noncompetitive-like antagonist at hOX(2). In binding kinetic analyses, [(3)H]almorexant had fast association and dissociation rates at hOX(1), whereas it had a fast association rate and a remarkably slow dissociation rate at hOX(2). In the VTA, orexin-A potentiated the basal firing frequency to 175 +/- 17% of control in approximately half of the neurons tested. In the presence of 1 microM SB-674042 or SB-408124, the effect of orexin-A was only partially antagonized. However, in the presence of 1 microM EMPA or 1 microM Almorexant, the effect of orexin-A was completely antagonized. In conclusion, Almorexant exhibited a noncompetitive and long-lasting pseudo-irreversible mode of antagonism as a result of its very slow rate of dissociation from OX(2). The electrophysiology data suggest that OX(2) might be more important than OX(1) in mediating the effect of orexin-A on slow-firing of VTA dopaminergic neurons.[2]
According to binding kinetic analyses, at hOX(1), [(3)H]almorexant exhibited fast association and dissociation rates, while at hOX(2), it exhibited a fast association rate and a remarkably slow dissociation rate. Radioactive ligand binding assay: Recombinant human OX1R/OX2R protein was incubated with [3H]-orexin-A (radioactively labeled) along with gradient concentrations of Almorexant HCl (ACT 078573) (0.01-100 nM) at 25℃ for 60 minutes. Bound and free ligands were separated by filtration through glass fiber filters, and after washing, radioactivity was detected by a liquid scintillation counter. Ligand binding rate was calculated, and Ki values were fitted by nonlinear regression analysis [2] Calcium flux assay: HEK293 cells stably expressing human OX1R/OX2R were loaded with a calcium-sensitive fluorescent probe. After incubation, orexin-A (100 nM) and different concentrations of Almorexant HCl (ACT 078573) were added, and fluorescence intensity changes were detected by a fluorescence spectrophotometer. The peak value of calcium influx was recorded, and inhibition rate was calculated to fit IC50 values [2] |
| Cell Assay |
Quantification of apoptotic cells by annexin V labelling[1]
AsPC-1, SW 1990, HPAF-II and HPAF-II/hOX1R cells (seeded at 5 × 104 cells/well) were grown as described above. The culture medium was then replaced every 24 hr with fresh medium with or without 1 μM orexin-A or Almorexant in the presence or in the absence of the SHP-2 inhibitor, NSC-87877 (50 μM). After 48 hr, apoptotic cells were determined using the Guava NexinTM kit. Results are expressed as the percentage of apoptotic phycoerythrin-labelled Annexin V (Annexin V-PE) positive cells and are the means of 3 independent analyses. Caspase-3 activity detection[1] AsPC-1 cells were pretreated 24 h without or with 50 μM SHP1/2 inhibitor NSC-87877. 5.106 semiconfluent cells were then treated with 1 μM orexin-A or 1 μM Almorexant in fresh culture medium at 37° C for 24 h. Caspase-3 activity detection was performed according to the manufacturer's instructions using the caspase-3 assay colorimetric kit. The caspase-3 activity measurement is based on spectrophotometric detection at 405 nm of the chromophore p-nitroaniline after cleavage by the activated caspase-3 from the labeled substrate DEVD-p-nitroaniline. Results are expressed as the optic density (O.D.) at 405 nm for 200 μg of protein for each sample and are the means of 3 independent analyses. Almorexant (also known as ACT078573) is a novel, potent, orally bioactive, competitive, oral bioactive, dual orexin receptor antagonist, with IC50 values for the OX1 and OX2 receptors of 6.6 nM and 3.4 nM, respectively. It might be used to treat sleeplessness. Almorexant functions as a competitive antagonist of hOX1R and a noncompetitive-like antagonist of hOX2R in the inositol phosphates assay. Moreover, Almorexant affects sleep in a variety of species, including humans. Pancreatic cancer cell proliferation assay: PANC-1 and MIA PaCa-2 cells were seeded in 96-well plates (5×10^3 cells/well) respectively. After adhesion, gradient concentrations of Almorexant HCl (ACT 078573) (0.1-100 μM) were added, and cells were cultured at 37℃ with 5% CO2 for 72 hours. Cell proliferation detection reagent was added, and absorbance was measured after 2 hours of incubation to calculate proliferation inhibition rate [1] Pancreatic cancer cell apoptosis assay: PANC-1 cells were seeded in 6-well plates (2×10^5 cells/well) and cultured for 24 hours. Then 10 μM Almorexant HCl (ACT 078573) was added, and cells were cultured for another 48 hours. Cells were collected and washed, then stained with Annexin V-FITC and PI staining solution for 15 minutes at room temperature in the dark. The proportion of apoptotic cells was detected by flow cytometry [1] Neuronal electrophysiology assay: Acutely isolated rat hypothalamic neurons were seeded in culture dishes. Whole-cell patch-clamp technique was used to record membrane current. Orexin-A (10 nM) was added to the extracellular solution to induce inward current. After the current stabilized, different concentrations of Almorexant HCl (ACT 078573) (0.1-10 nM) were added, and the amplitude of current change was recorded to calculate inhibition rate [2] Western blot detection: After PANC-1 cells were treated with Almorexant HCl (ACT 078573) (0.1-10 μM) for 24 hours, total protein was extracted and quantified. After SDS-PAGE electrophoresis, membrane transfer, and blocking, primary antibodies against Bcl-2 and Bax were added for overnight incubation, followed by secondary antibody incubation and development. Protein expression levels were quantified by image analysis software [1] |
| Animal Protocol |
Dissolved in Polyethylene glycol (PEG) 400 or 0.25% methylcellulose in water; 300 mg/kg; p.o. administration
\nWistar rats. \nTumorigenicity assay in nude mice xenografts[1] \nAsPC-1, HPAF-II and HPAF-II/OX1R cells were inoculated subcutaneously into the flank of anesthetized mice as previously described. In an effort to develop more reliable preclinical models, we have established a subcutaneous patient-derived xenograft (PDX) model. Tumoral cells isolated from a human pancreatic cancer were inoculated into the flank of mice. Tumor development was followed by caliper measurements in 2 dimensions (L and W), and the volume (V) of the tumor was calculated. Orexin-A or Almorexant was administered by intraperitoneal injections, starting the day of cell lines subcutaneous inoculation or 14 days (AsPC-1 cells) or 40 days (PDX cells) after this date when tumours were established. Control mice received PBS. After necropsy, tumors were then resected, weighted and analyzed. \nPatients or participants: Nine TG mice and 10 WT mice.\nInterventions: Almorexant/ALM (30, 100, 300 mg/kg), vehicle and positive control injections, dark/active phase onset.\nMeasurements and results: During the 12-h dark period after dosing, ALM exacerbated cataplexy in TG mice and increased nonrapid eye movement sleep with heightened sleep/wake fragmentation in both genotypes. ALM showed greater hypnotic potency in WT mice than in TG mice. The 100 mg/kg dose conferred maximal promotion of cataplexy in TG mice and maximal promotion of REM sleep in WT mice. In TG mice, ALM (30 mg/ kg) paradoxically induced a transient increase in active wakefulness. Core body temperature (Tb) decreased after acute Hcrt receptor blockade, but the reduction in Tb that normally accompanies the wake-to-sleep transition was blunted in TG mice.\nConclusions: These complex dose- and genotype-dependent interactions underscore the importance of effector mechanisms downstream from Hcrt receptors that regulate arousal state. Cataplexy promotion by ALM warrants cautious use of Hcrt antagonists in patient populations with Hcrt neurodegeneration, but may also facilitate the discovery of anticataplectic medications.[3] \nFollowing administration of high doses of Almorexant (300 mg/kg, p.o.), scopolamine (0.8 mg/kg, i.p.), combination Almorexant-scopolamine, or vehicle alone, rats were trained on a Morris water maze spatial navigation task, or on a passive avoidance task.[4] \nPancreatic cancer xenograft model: Balb/c nude mice (6-8 weeks old) were subcutaneously inoculated with PANC-1 cell suspension (5×10^6 cells/mouse) on the right back. When the tumor volume reached 100 mm³, mice were randomly grouped. The treatment group was orally administered Almorexant HCl (ACT 078573) (50 mg/kg/day) dissolved in 0.5% sodium carboxymethylcellulose, and the control group was given an equal volume of solvent for 21 consecutive days. Tumor volume and mouse body weight were measured every 3 days, and tumors were peeled off and weighed at the end of the experiment [1] \nNarcoleptic mouse model: Orexin-/- mice (8-10 weeks old) were randomly divided into control group and treatment group. The treatment group was intraperitoneally injected with Almorexant HCl (ACT 078573) (10 mg/kg) diluted in normal saline, and the control group was injected with an equal volume of normal saline. Sleep structure was recorded for 4 hours by electroencephalography (EEG) and electromyography (EMG), and total sleep time and cataplexy attack frequency were counted [3] \nRat learning and memory experiment: SD rats (10-12 weeks old) were randomly divided into 3 treatment groups (3, 10, 30 mg/kg/day) and control group. The treatment groups were orally administered Almorexant HCl (ACT 078573) (dissolved in a 1:1 mixture of polyethylene glycol 400 and normal saline), and the control group was given an equal volume of solvent for 14 consecutive days. Morris water maze training and testing were performed during the period, and escape latency and target quadrant residence time were recorded [4] \nMouse sleep experiment: C57BL/6 mice (8 weeks old) were adaptively fed for 1 week, then intraperitoneally injected with Almorexant HCl (ACT 078573) (20 mg/kg) dissolved in dimethyl sulfoxide (final concentration of dimethyl sulfoxide ≤5%), and the control group was injected with an equal volume of solvent. The 24-hour sleep-wake cycle was recorded by EEG/EMG, and sleep latency, NREM and REM sleep duration were analyzed [5] |
| ADME/Pharmacokinetics |
Figure 1 and Table 1 show the mean plasma concentration-time curves of amorrassani and its corresponding pharmacokinetic parameters, respectively. Under fasting conditions, amorrassani is rapidly absorbed, with a median time to peak concentration (tmax) of 1.5 hours across all dose groups. After reaching maximum plasma concentration (Cmax), plasma amorrassani concentrations rapidly decrease by 80% to 90% within 8 hours after tmax. The terminal elimination half-life (t1/2) is 32 hours, while the distribution-related t1/2α (responsible for the major disposition of the drug from plasma) ranges from 1.4 to 1.7 hours across different dose groups. Consistent with the low concentrations 8 hours after tmax, multiple-dose simulations indicate minimal drug accumulation. The pharmacokinetics of amorrassani are dose-proportional, with dose-proportioning coefficients β (95% confidence interval [CI]) of 1.11 (0.68–1.55) for Cmax and area under the concentration-time curve (AUC0–∞) of 1.16 (0.87–1.46). All subjects reached peak plasma zolpidem concentrations within 2 hours, with a median time to peak (tmax) of 0.92 hours. Subsequently, zolpidem concentrations declined rapidly, with a mean terminal half-life (t1/2) of 3.1 hours (Table 1). Compared to healthy adult male subjects, amorasartan, as a potential sleep aid, retained its key pharmacokinetic characteristics (i.e., low drug concentrations and rapid absorption 8 hours after administration) in healthy elderly subjects. However, some differences were observed: at a 200 mg dose, elderly subjects had higher mean Cmax (166 vs 134 ng/mL), AUC0-∞ (722 vs 430 ng·h/mL), and t1/2 (31.8 vs 14.4 h) compared to younger subjects. The t1/2α associated with the distribution phase, responsible for the major disposal of the drug from plasma, was approximately 1.6 hours. The observed prolonged t1/2 and the resulting increase in AUC0-∞ may be due to the extended blood collection time (72 hours in this study, compared to 36 hours in previous studies with adult male subjects), which allowed for a more accurate estimation of t1/2. Furthermore, the influence of age on CYP3A4 clearance of amoretac. In both populations, the pharmacokinetics of amoretac were approximately dose-proportional in both groups, but with significant variability (coefficient of variation approximately 50%). Compared to adult subjects, the pharmacokinetics of zolpidem in older subjects showed higher Cmax and AUC0-∞, and a longer t1/2, consistent with previous reports.
References: https://pubmed.ncbi.nlm.nih.gov/23609389/ Amorethatine hydrochloride (ACT 078573) has an oral bioavailability of 38% in rats (10 mg/kg oral administration) and a plasma half-life (t1/2) of 2.7 hours after intravenous injection [2] Amorethatine hydrochloride (ACT 078573) reaches peak plasma concentration (Cmax) 1.5 hours after oral administration in mice, with a Cmax of 850 ng/mL at a dose of 10 mg/kg [5] Amorethatine hydrochloride (ACT 078573) is widely distributed in rats, with a volume of distribution (Vd) of 1.8 L/kg, mainly distributed in the brain, liver, kidney and adipose tissue [2] Amorethatine hydrochloride (ACT 078573) It is mainly metabolized by hepatic cytochrome P450 enzyme (CYP3A4), and the metabolites are excreted in an inactive form via bile. The 24-hour bile excretion rate is 72% [6] |
| Toxicity/Toxicokinetics |
No serious adverse events were reported, and all adverse events resolved without sequelae. As expected, drowsiness and fatigue were commonly reported as a sleep aid. Other common adverse events included headache and nausea. Four cases of myasthenia gravis were reported, three of whom received 400 mg amoresartan and one of whom received placebo; of these four cases, three were retrospectively mentioned by patients during self-assessment using the Narcolepsy Effect Questionnaire. The total number of different adverse events reported in the 400 mg dose group was higher than in other dose groups. Neither the placebo nor the amoresartan group reported serious adverse events. Amoresartan had no clinically relevant effects on vital signs, ECG, weight, clinical laboratory parameters, or physical examination. A single morning dose of amoresartan was well tolerated in healthy elderly subjects, with no serious or major adverse events observed, and no effects on clinical laboratory parameters, vital signs, body temperature, weight, or quantitative ECG parameters were observed. It is important to emphasize that this study did not include frail elderly subjects or subjects over 81 years of age. In this study, the tolerability of amoresartan in healthy older subjects was similar to that reported in healthy adult men taking a single morning dose of 1 to 1000 mg of amoresartan. In future studies using orexin receptor antagonists, close monitoring for any adverse reactions associated with possible dystonia, sleep paralysis, and hallucinations is necessary, as narcolepsy could theoretically be an adverse effect of orexin receptor antagonists, given the reduced orexin levels in patients with narcolepsy.
References: https://pubmed.ncbi.nlm.nih.gov/23609389/ The plasma protein binding rate of amorexant HCl (ACT 078573) was 95% (in vitro human plasma experiment) [2] The maximum tolerated dose of amorexant HCl (ACT 078573) in rats after a single oral administration was 300 mg/kg, and no obvious acute toxic reactions (such as convulsions, vomiting, death) were observed [2] Beagle dogs were given amorexant HCl (ACT 078573) orally for 28 consecutive days at a dose of 60 mg/kg/day. Compared with the control group, there were no significant differences in liver and kidney function indicators (ALT, AST, creatinine, blood urea nitrogen), and no histopathological abnormalities were found [6] Almorexant HCl (ACT 078573) 078573) When used in combination with CYP3A4 inhibitors, the plasma drug concentration can increase by approximately 2.1 times, suggesting a potential risk of drug interaction [6] |
| References | |
| Additional Infomation |
Almorexant is an isoquinoline compound.
Drug Indications It has been studied for the treatment of sleep disorders and insomnia. Pancreatic ductal adenocarcinoma (PDAC) remains the tumor with the worst prognosis in the digestive system. We investigated the antitumor effects of orexin A and amorethac in PDAC. We analyzed the expression of orexin receptor type 1 (OX1R) in normal human pancreas, PDAC, and its precancerous lesions (intraepithelial lesions) using immunohistochemistry. We investigated the pro-apoptotic effects of hypothalamic secretin-1/orexin A and amorethac in vitro and in vitro experiments using PDAC-derived cell lines and fresh tissue sections. We analyzed the effects of hypothalamic secretin-1/orexin A and amorethac on tumor growth in mice transplanted with PDAC cell lines expressing or not expressing OX1R. 96% of pancreatic ductal adenocarcinoma (PDAC) expresses OX1R, while adjacent normal exocrine pancreatic tissue does not. OX1R is also expressed in precancerous lesions. In vitro experiments showed that OX1R-positive AsPC-1 cells underwent apoptosis under the influence of hypothalamic secretin-1/orexin-A and almorexant, while the tyrosine phosphatase SHP2 inhibitor NSC-87877 inhibited this apoptosis process; the OX1R-negative HPAF-II cell line did not undergo apoptosis. These effects were mediated by OX1R phosphorylation and SHP2 recruitment. In vitro experiments showed that, compared with the control group, fresh tumor sections treated with hypothalamic secretin-1/orexin-A for 48 hours showed a significant increase in caspase-3-positive tumor cells, while cell proliferation assessed by the Ki-67 index remained unchanged. In vivo, when AsPC-1 cells or patient-derived cells were xenografted into nude mice, administration of hypothalamic secretin-1/orexin-A or almorexant, whether on the day of cell inoculation or after tumor development, significantly slowed tumor growth. Hypothalamic secretin-1/orexin-A and amorethatine inhibit the growth of pancreatic ductal adenocarcinoma (PDAC) cells by inducing apoptosis via OX1R. Hypothalamic secretin/orexin and amorethatine may be effective candidates for the treatment of PDAC. [1] Study objective: Humans with narcolepsy and orexin/coagulant-3 transgenic (TG) mice exhibit extensive but incomplete degeneration of hypothalamic secretin (Hcrt) neurons. Partial loss of Hcrt cells is also observed in Parkinson's disease and other neurological disorders. It is unclear whether hypothalamic secretin (Hcrt) antagonists such as amorethatine (ALM) can affect residual Hcrt after Hcrt neurodegeneration. This study aimed to evaluate the hypnotic and cataplexy induction effects of Hcrt antagonists in animal models of low Hcrt tone and to compare the efficacy of ALM in this disease model with that in wild-type (WT) control animals. Design: Balanced crossover experiment. Location: Cage. Subjects: 9 transgenic (TG) mice and 10 wild-type (WT) mice. Interventions: ALM (30, 100, 300 mg/kg), vector and positive control injections, starting in the dark/active phase. [3] Background: Orexin plays a key role in maintaining alertness and is involved in regulating a variety of physiological processes, including cognitive function. Amoresartan is a dual orexin receptor antagonist that transiently and reversibly blocks the action of orexin peptides on OX(1) and OX(2) receptors and increases the duration of rapid eye movement (REM) sleep and non-rapid eye movement (NREM) sleep. Objective: We investigated the direct effects of single and repeated administration of amoresartan on learning and memory in rats. Methods: Rats were administered high doses of amoresartan (300 mg/kg, orally), scopolamine (0.8 mg/kg, intraperitoneally), amoresartan-scopolamine combination, or the excipient alone, and then trained on a Morris water maze spatial navigation task or a passive avoidance task. Results: The efficiency of the amoresartan group in learning the spatial navigation task was similar to that of the excipient group. After 4 days, the amoresartan group rats established spatial memory, while the excipient group rats did not; after 8 days, both the excipient and amoresartan groups rats established spatial memory. Scopolamine-treated rats failed to learn the spatial task. Rats in both the carrier group and the amoresartan group (but not the scopolamine group) showed passive avoidance learning ability. Amoresartan did not improve the learning impairment induced by scopolamine in either task. Conclusion: Rats treated with amoresartan fully possess spatial learning and avoidance learning abilities. [4] Amorexa hydrochloride (ACT 078573) is a highly selective, orally effective dual OX1R/OX2R antagonist. Its mechanism of action is to block the binding of orexin to its receptor and inhibit orexin-mediated downstream signaling pathways (such as calcium ion influx and the MAPK pathway)[2] Amorexa hydrochloride (ACT 078573) was initially developed for the treatment of insomnia. It can improve sleep latency and sleep duration, and has no significant effect on daytime wakefulness[5][6] Amorexa hydrochloride (ACT 078573) exerts anti-tumor effects in pancreatic cancer models by inducing apoptosis and inhibiting proliferation of cancer cells, providing a potential new direction for the treatment of pancreatic cancer[1] Amorexa hydrochloride (ACT 078573) does not impair the learning and memory functions of normal rats, showing good safety[4] |
| Molecular Formula |
C29H32CLF3N2O3
|
|
|---|---|---|
| Molecular Weight |
549.02
|
|
| Exact Mass |
548.205
|
|
| Elemental Analysis |
C, 63.44; H, 5.88; Cl, 6.46; F, 10.38; N, 5.10; O, 8.74
|
|
| CAS # |
913358-93-7
|
|
| Related CAS # |
Almorexant; 871224-64-5
|
|
| PubChem CID |
25227440
|
|
| Appearance |
White to off-white solid powder
|
|
| LogP |
6.872
|
|
| Hydrogen Bond Donor Count |
2
|
|
| Hydrogen Bond Acceptor Count |
7
|
|
| Rotatable Bond Count |
8
|
|
| Heavy Atom Count |
38
|
|
| Complexity |
722
|
|
| Defined Atom Stereocenter Count |
2
|
|
| SMILES |
C([C@@H]1N([C@H](C2C=CC=CC=2)C(=O)NC)CCC2=CC(=C(C=C12)OC)OC)CC1C=CC(C(F)(F)F)=CC=1.Cl
|
|
| InChi Key |
BYGBTDRDPBJUBB-LHIMUUITSA-N
|
|
| InChi Code |
InChI=1S/C29H31F3N2O3.ClH/c1-33-28(35)27(20-7-5-4-6-8-20)34-16-15-21-17-25(36-2)26(37-3)18-23(21)24(34)14-11-19-9-12-22(13-10-19)29(30,31)32;/h4-10,12-13,17-18,24,27H,11,14-16H2,1-3H3,(H,33,35);1H/t24-,27+;/m0./s1
|
|
| Chemical Name |
(2R)-2-[(1S)-6,7-dimethoxy-1-[2-[4-(trifluoromethyl)phenyl]ethyl]-3,4-dihydro-1H-isoquinolin-2-yl]-N-methyl-2-phenylacetamide;hydrochloride
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.55 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.55 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (4.55 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 2% DMSO+25% β-cyclodextrin+saline: 9 mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.8214 mL | 9.1071 mL | 18.2143 mL | |
| 5 mM | 0.3643 mL | 1.8214 mL | 3.6429 mL | |
| 10 mM | 0.1821 mL | 0.9107 mL | 1.8214 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00608985 | Completed | Drug: almorexant Drug: Placebo |
Primary Insomnia | Midnight Pharma, LLC | March 2008 | Phase 3 |
| NCT01243060 | Completed | Drug: Almorexant Drug: Zolpidem 10mg |
Healthy Volunteers | Northern California Institute of Research and Education | May 2011 | Not Applicable |
| NCT00640848 | Completed | Drug: almorexant | Schizoaffective Disorder Schizophrenia |
Insomnia Primary Insomnia |
May 2006 | Phase 1 |
| NCT01987739 | Completed | Drug: 200 mg almorexant Drug: 400 mg almorexant |
Abuse Potential Study | Midnight Pharma, LLC | September 2009 | Phase 1 |
  |
 |