
| Size | Price | Stock | Qty |
|---|---|---|---|
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
|
Purity: =99.95%
Palbociclib (formerly PD-0332991; PD0332991; Pfizer trade name Ibrance) is a highly selective, orally bioavailable pyridopyrimidine-based CDK4/6 inhibitor that has been approved for cancer treatment. In cell-free experiments, it inhibits CDK4/6 with an IC50 of 11 nM and 16 nM, respectively. Many tumor cells overexpress CDK4 and CDK6, and Pfizer's palbociclib is the first CDK4/6 inhibitor to be approved by the FDA as a cancer treatment in 2017. There is no evidence of any activity against PDGFR, EGFR, FGFR, CDK1/2/5, InsR, etc. In vitro, it is a strong anti-proliferative agent that induces an exclusive G1 arrest in Rb-positive tumor cells. It has been shown to cause G1 arrest in primary bone marrow cells and stop tumor growth in disseminated human myeloma xenografts.
| Targets |
Cdk4/cyclin D3 (IC50 = 9 nM); Cdk4/cyclin D1 (IC50 = 11 nM); Cdk6/cyclin D2 (IC50 = 16 nM); DYRK1A (IC50 = 2000 nM); MAPK (IC50 = 8000 nM)
|
|---|---|
| ln Vitro |
Palbociclib is a CKD4/6 inhibitor, dramatically lowers the viability of HR+/HER2+ tumor cells when combined with fulvestrant and tucatinib. We next investigated potential cross-resistance mechanisms between two key components of the triple combination: tucatinib and palbociclib. We generated TR and palbociclib-resistant (PR) subclones of the BT474 and MDA-MB-361. We did not generate fulvestrant-resistant subclones, as all HR+/HER2+ cell lines exhibit intrinsic resistance to ER-targeting agents because of HER2 overexpression. We hypothesized that the mechanisms of resistance to tucatinib and palbociclib would be non-overlapping; therefore, cross-treatment of TR subclones with palbociclib and PR subclones with tucatinib would result in effective cancer cell killing.
In clonogenic assays, MDA-MB-361 TR and BT474 TR cells survived treatment with 0.67 μmol/L tucatinib; however, they had a significant reduction in clonogenic survival when treated with 3 μmol/L palbociclib (Fig. 3A and B). The addition of tucatinib to palbociclib did not improve reduction of clonogenic survival, reflecting profound resistance of TR subclones to tucatinib.[2]
PD 0332991 is a highly specific inhibitor of cyclin-dependent kinase 4 (Cdk4) (IC50, 0.011 micromol/L) and Cdk6 (IC50, 0.016 micromol/L), having no activity against a panel of 36 additional protein kinases. It is a potent antiproliferative agent against retinoblastoma (Rb)-positive tumor cells in vitro, inducing an exclusive G1 arrest, with a concomitant reduction of phospho-Ser780/Ser795 on the Rb protein [1]. |
| ln Vivo |
Oral administration of PD 0332991 to mice bearing the Colo-205 human colon carcinoma produces marked tumor regression. Therapeutic doses of PD 0332991 cause elimination of phospho-Rb and the proliferative marker Ki-67 in tumor tissue and down-regulation of genes under the transcriptional control of E2F. The results indicate that inhibition of Cdk4/6 alone is sufficient to cause tumor regression and a net reduction in tumor burden in some tumors.[1]
Palbociclib a CKD4/6 inhibitor, exhibits superior efficacy in suppressing tumor growth in vivo when combinates with tucatinib and fulvestrant.[2] Triple combination shows superior efficacy in suppression of tumor growth in vivo [2] Researchers next tested our hypothesis that the combination of tucatinib, palbociclib, and fulvestrant would have efficacy in vivo. For in vivo experiments, we focused our analysis on the triple combination and dual combinations containing tucatinib (tucatinib plus fulvestrant or tucatinib plus palbociclib) and compared their activity with vehicle and single-agent tucatinib. We have elected not to test activity of single agents palbociclib, fulvestrant, or a combination of palbociclib and fulvestrant in animal experiments because published clinical studies have shown that HER2 inhibition is critical for treatment of patients with HER2+ breast cancer; the addition of HER2 inhibitors to combination treatment regimens improves OS (41, 42). Therefore, only tucatinib containing combinations are clinically relevant for patients with HR+/HER2+ disease. MDA-MB-361 tumor growth was not reduced by tucatinib compared with vehicle (Fig. 2A). However, the combination of tucatinib with fulvestrant or palbociclib markedly reduced tumor growth, and the triple combination induced the most robust reduction in tumor growth. Average tumor volume at the EOT (VEOT) did not differ between tucatinib and vehicle groups, whereas VEOT in tucatinib plus palbociclib, tucatinib plus fulvestrant, and the triple combination groups were significantly smaller (Fig. 2B and C). VEOT on triple combination therapy was significantly reduced compared with both dual combinations (Fig. 2B and C). There was no difference in average TGR between vehicle and tucatinib; however, both dual combinations and the triple combination significantly reduced TGR (Fig. 2D). The TGR was the lowest in the triple combination, approximately 5.6 times lower than vehicle (Fig. 2D). IHC staining of Ki67 revealed that all palbociclib containing combinations had marked reduction of proliferation rate. However, the lowest percentage of proliferating cells was observed in the triple combination, where Ki67 was significantly lower compared with the best dual combination (Fig. 2E and F). BT474 tumors yielded similar results where the triple combination induced the most robust reduction in tumor growth (Fig. 2G). Tucatinib alone or dual combination with fulvestrant or palbociclib also reduced tumor growth compared with vehicle treatments, although to a lesser extent than triple combination. Average VEOT in triple combination was significantly lower compared with tucatinib alone, tucatinib plus fulvestrant, or tucatinib plus palbociclib groups (Fig. 2H and I). The triple combination therapy was the only treatment where tumors were regressed in size as compared with the start of experiment. The triple combination induced a negative TGR, substantially lower than the TGR in the groups treated with single-agent tucatinib or tucatinib plus fulvestrant (Fig. 2J). Finally, Ki67 analysis revealed that all palbociclib containing combinations had marked reduction of proliferation rate, with the lowest Ki67 in the groups treated with palbociclib plus tucatinib or the triple combination (Fig. 2K and L). Full statistical analysis of animal experiments is summarized (Supplementary Fig. S4A). Mice did not experience significant weight loss on any treatment regimen, suggesting low toxicity of the triple combination in mammals (Supplementary Fig. S4B and S4C). |
| Enzyme Assay |
In DMSO, a stock solution of PD0332991 is made. CDK assays are run on filter plates with 96 wells. By infecting insect cells with baculovirus, all CDK-cyclin kinase complexes are expressed and purified. A portion of pRb fused to GST (GST·RB-Cterm) spanning amino acids 792–928 serves as the substrate for the assays. Each well has a total volume of 0.1 mL and contains the following final concentrations: 20 mM Tris-HCl, pH 7.4, 50 mM NaCl, 1 mM dithiothreitol, 10 mM MgCl2, 25 μM ATP (for CDK4-cyclin D1, CDK6-cyclin D2, and CDK6-cyclin D3), or 12 μM ATP (for CDK2-cyclin E, CDK2-cyclin A, and CDC2-cyclin B). This mixture also contains 0.25 μCi of [γ-32P]ATP, 20 ng of enzyme, 1 μg of GST·RB-Cterm, and PD 0332991 (0.001-0.1μM). The plate is placed on a plate mixer for two minutes after all ingredients—aside from the [γ-32P]ATP—are added to the wells. The plate is incubated at 25°C for 15 minutes after the addition of [γ-32P]ATP to initiate the reaction. The plate is kept at 4 °C for at least an hour to allow the substrate to precipitate before the reaction is stopped by adding 0.1 mL of 20% trichloroacetic acid. Next, 0.2 mL of 10% trichloroacetic acid is used to wash the wells five times, and a β plate counter is used to measure the radioactive incorporation.
|
| Cell Assay |
Cell viability is assessed using the Cell Titer Glo assay following a 72-hour vehicle or medication treatment. Prior to fulvestrant treatment, cells are grown in conditions free of estrogen and estradiol is added to a final concentration of 10-8 M. For every cell line, the IC30 values for palbociclib, fulvestrant, and tucatinib are determined; the IC30 concentrations are then utilized in the ensuing studies.
|
| Animal Protocol |
NCG mice injected with MDA-MB-361 cells
50mg/kg o.g. Oral administration of PD 0332991 to mice bearing the Colo-205 human colon carcinoma produces marked tumor regression. Therapeutic doses of PD 0332991 cause elimination of phospho-Rb and the proliferative marker Ki-67 in tumor tissue and down-regulation of genes under the transcriptional control of E2F. The results indicate that inhibition of Cdk4/6 alone is sufficient to cause tumor regression and a net reduction in tumor burden in some tumors.[1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion
Palbociclib presents a linear pharmacokinetic profile and its peak plasma concentration was observed 6-12 hours after oral administration. The oral bioavailability is reported to be of 46% with a steady-state reached after 8 days and a median accumulation ratio of 2.4. The absorption of palbociclib is significantly reduced under fasting conditions and hence, food intake is recommended when this drug is administered. The main route of elimination of palbociclib is through feces after hepatic metabolism while renal clearance seems to play a minor role accounting only for 17.5% of the eliminated dose. The mean apparent distribution of palbociclib is 2583 L which suggests that palbociclib penetrates extensively into peripheral tissues. The mean apparent oral clearance of palbociclib is of 63.1 L/h. Metabolism / Metabolites Palbociclib is mainly hepatically transformed. the metabolism is mainly performed by the activities of the cytochrome P450 isoenzyme 3A and the sulfotransferase 2A1. The metabolism of palbociclib is represented mainly by reactions of oxidation and sulfonation followed by acylation and glucuronidation as minor reactions. After its metabolism, palbociclib forms mainly inactive glucuronide and sulfamic acid conjugates. The major circulating metabolite, accounting for 1.5% of the dose in excreta is is the glucuronide conjugate. Biological Half-Life The mean plasma elimination half-life of palbociclib is 29 hours. |
| Toxicity/Toxicokinetics |
Hepatotoxicity
In the large clinical trials, adverse events were common and led to dose reductions in one-third of patients and discontinuation in 8%. Publications on the efficacy and safety of palbociclib rarely mentioned serum ALT elevations or hepatotoxicity. In a study of women with refractory, metastatic breast cancer, serum ALT elevations occurred in 6% [2% over 5 times ULN] receiving palbociclib and fulvestrant compared to 3% [none over 5 times ULN] on fulvestrant alone. Since its approval and more widescale use, there have been several reports of prominent ALT elevations arising after 2 or 3 cycles of palbociclib, that improved on discontinuation and recurred rapidly when restarted. Serum bilirubin and alkaline phosphatase levels were normal and symptoms were not mentioned. In addition, there have been rare reports of patients with refractory metastatic breast cancer who developed pseudocirrhosis within 2 to 3 months of starting palbociclib presenting with fatigue, jaundice and ascites with only modest elevations in serum aminotransferase and alkaline phosphatase levels. Imaging revealed a severely nodular liver, but liver histology showed desmoplastic changes in areas of necrotic metastatic tumor without cirrhosis. The liver also had vascular changes suggestive of sinusoidal obstruction syndrome, changes possibly caused by the dramatic involution of the metastatic tumor tissue combined with vascular damage. Pseudocirrhosis has been reported with other highly successful antineoplastic therapies of cancer metastatic to the liver, but the frequency is rare. Likelihood score: C (probable rare cause of clinically apparent liver injury that may represent pseudocirrhosis from nodular transformation of the liver in response to necrosis of hepatic metastases). Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation No information is available on the clinical use of palbociclib during breastfeeding. Because palbociclib is 85% bound to plasma proteins, the amount in milk is likely to be low. However, its half-life is about 29 hours and it might accumulate in the infant. It is also given in combination with letrozole or fulvestrant, which may increase the risk to the infant. The manufacturer recommends that breastfeeding be discontinued during palbociclib therapy and for 3 weeks after the last dose. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Binding of palbociclib to human plasma proteins in vitro accounts for approximately 85% of the administered dose. |
| References | |
| Additional Infomation |
Pharmacodynamics
Due to its mechanism of action, palbociclib inhibits cell growth and suppresses DNA replication in retinoblastoma tumor suppressor gene (RB) proficient cancer cells. As expected, these RB cells present a significant increase in the proportion of cells in G1 state and the presence of palbociclib produces effective dephosphorylation of RB, reduce proliferation and induce senescence causing cell-cycle arrest. In vitro studies showed the potential for palbociclib to reduce cellular proliferation of estrogen receptor-positive breast cancer cell lines through the inhibition of the cell-cycle progression from G1 to S phase. In this study, it was demonstrated that the sensitivity of the cells significantly increased with the expression of _RB1_ and _CCND1_ and low expression of _CDKN2A_. As well, palbociclib, combined with antiestrogens, enhanced _in vivo_ antitumor activity in estrogen receptor-positive breast cancer mouse models. In clinical trials, palbociclib, in combination with letrozole, was shown to significantly increase the progression-free survival (PFS) in patients with metastatic breast cancer without prior endocrine treatment. In the results, the PFS increased from 4.5 to 9.5 months with an overall response rate (ORR) of 24.6%. Breast cancers that express hormonal receptors (HR) and HER2 display resistance to targeted therapy. Tumor-promotional signaling from the HER2 and estrogen receptor (ER) pathways converges at the cyclin D1 and cyclin-dependent kinases (CDK) 4 and 6 complex, which drives cell-cycle progression and development of therapeutic resistance. Therefore, we hypothesized that co-targeting of ER, HER2, and CDK4/6 may result in improved tumoricidal activity and suppress drug-resistant subclones that arise on therapy. We tested the activity of the triple targeted combination therapy with tucatinib (HER2 small-molecule inhibitor), palbociclib (CKD4/6 inhibitor), and fulvestrant (selective ER degrader) in HR+/HER2+ human breast tumor cell lines and xenograft models. In addition, we evaluated whether triple targeted combination prevents growth of tucatinib or palbociclib-resistant subclones in vitro and in vivo. Triple targeted combination significantly reduced HR+/HER2+ tumor cell viability, clonogenic survival, and in vivo growth. Moreover, survival of HR+/HER2+ cells that were resistant to the third drug in the regimen was reduced by the other two drugs in combination. We propose that a targeted triple combination approach will be clinically effective in the treatment of otherwise drug-resistant tumors, inducing robust responses in patients.[2] |
| Molecular Formula |
C24H29N7O2
|
|---|---|
| Molecular Weight |
447.54
|
| Exact Mass |
447.238
|
| Elemental Analysis |
C, 64.41; H, 6.53; N, 21.91; O, 7.15
|
| CAS # |
571190-30-2
|
| Related CAS # |
Palbociclib monohydrochloride;827022-32-2;Palbociclib hydrochloride;571189-11-2;Palbociclib-d8;1628752-83-9;Palbociclib isethionate;827022-33-3;Palbociclib dihydrochloride;Palbociclib orotate;2757498-64-7;Palbociclib-d4 hydrochloride
|
| PubChem CID |
5330286
|
| Appearance |
Yellow solid powder
|
| Density |
1.3±0.1 g/cm3
|
| Boiling Point |
711.5±70.0 °C at 760 mmHg
|
| Melting Point |
200ºC
|
| Flash Point |
384.1±35.7 °C
|
| Vapour Pressure |
0.0±2.3 mmHg at 25°C
|
| Index of Refraction |
1.648
|
| LogP |
0.99
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
8
|
| Rotatable Bond Count |
5
|
| Heavy Atom Count |
33
|
| Complexity |
775
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
O=C1C(C(C([H])([H])[H])=O)=C(C([H])([H])[H])C2=C([H])N=C(N([H])C3C([H])=C([H])C(=C([H])N=3)N3C([H])([H])C([H])([H])N([H])C([H])([H])C3([H])[H])N=C2N1C1([H])C([H])([H])C([H])([H])C([H])([H])C1([H])[H]
|
| InChi Key |
AHJRHEGDXFFMBM-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C24H29N7O2/c1-15-19-14-27-24(28-20-8-7-18(13-26-20)30-11-9-25-10-12-30)29-22(19)31(17-5-3-4-6-17)23(33)21(15)16(2)32/h7-8,13-14,17,25H,3-6,9-12H2,1-2H3,(H,26,27,28,29)
|
| Chemical Name |
6-acetyl-8-cyclopentyl-5-methyl-2-[(5-piperazin-1-ylpyridin-2-yl)amino]pyrido[2,3-d]pyrimidin-7-one;hydrochloride
|
| Synonyms |
PD0332991; Palbociclib free base; UNII-G9ZF61LE7G; 6-acetyl-8-cyclopentyl-5-methyl-2-((5-(piperazin-1-yl)pyridin-2-yl)amino)pyrido[2,3-d]pyrimidin-7(8H)-one; PD-0332991; PD 0332991; Trade name: Ibrance.
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2 mg/mL (4.47 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2 mg/mL (4.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: 6.67 mg/mL (14.90 mM) in 0.5% CMC/saline water (add these co-solvents sequentially from left to right, and one by one), suspension solution; Need ultrasonic and warming and heat to 42°C. |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2344 mL | 11.1722 mL | 22.3444 mL | |
| 5 mM | 0.4469 mL | 2.2344 mL | 4.4689 mL | |
| 10 mM | 0.2234 mL | 1.1172 mL | 2.2344 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Sotorasib Activity in Subjects With Advanced Solid Tumors With KRAS p.G12C Mutation (CodeBreak 101)
CTID: NCT04185883
Phase: Phase 1 Status: Recruiting
Date: 2024-11-29
Evaluation of IC50concentrations of the CDK inhibitors dinaciclib and palbociclib on proliferation, and their effects on CDK-Rb-E2F signaling in human HPASMCs from healthy donors and IPAH patients.Nat Commun.2019May 17;10(1):2204. |
|---|
Effects of the CDK inhibitors dinaciclib and palbociclib on proliferation, cell cycle, and apoptosis.Nat Commun.2019May 17;10(1):2204. |
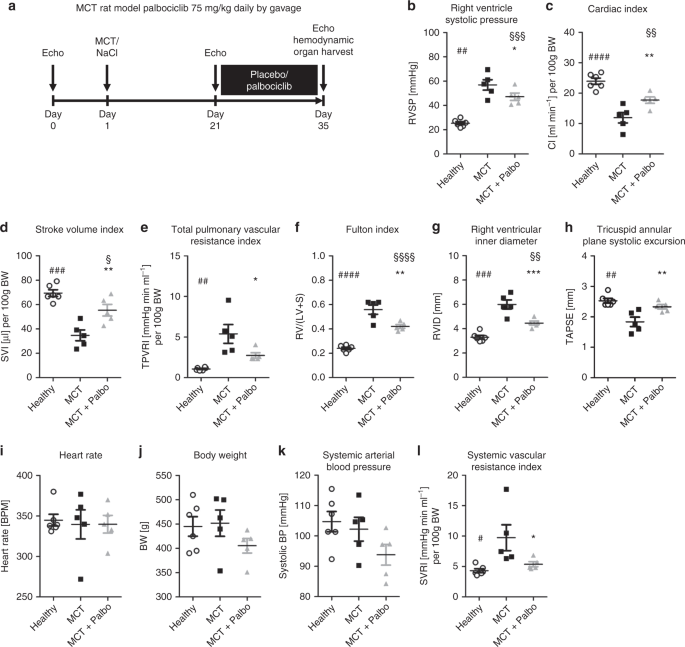 Effects of palbociclib on disease progression in the MCT rat model of pulmonary arterial hypertension.Nat Commun.2019May 17;10(1):2204. |
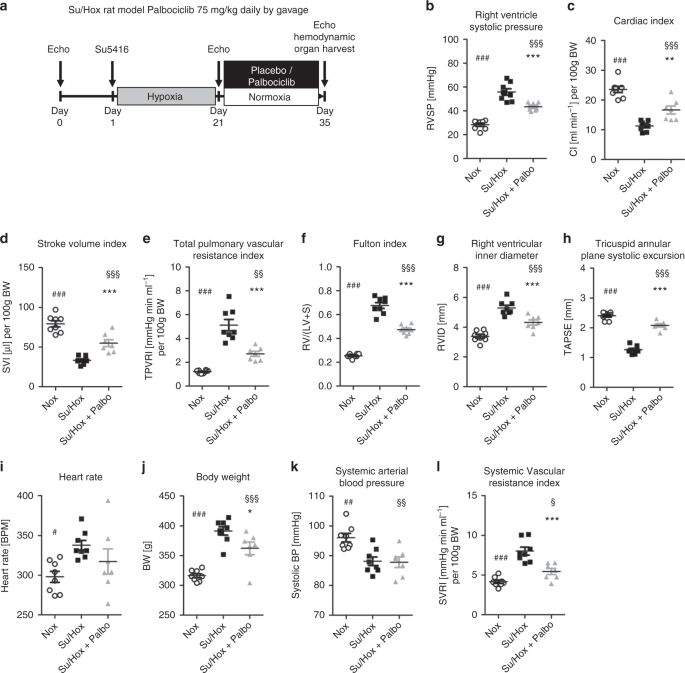 Effects of palbociclib on disease progression in the Su/Hox rat model of pulmonary arterial hypertension.Nat Commun.2019May 17;10(1):2204. |
|---|
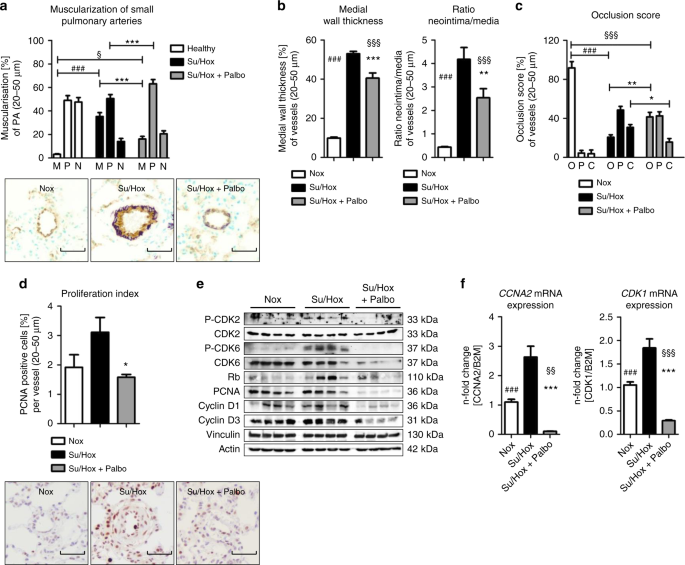 Ex vivo analyses of lung tissue for reversal of remodeling and in vivo drug efficacy in the Su/Hox model.Nat Commun.2019May 17;10(1):2204. |
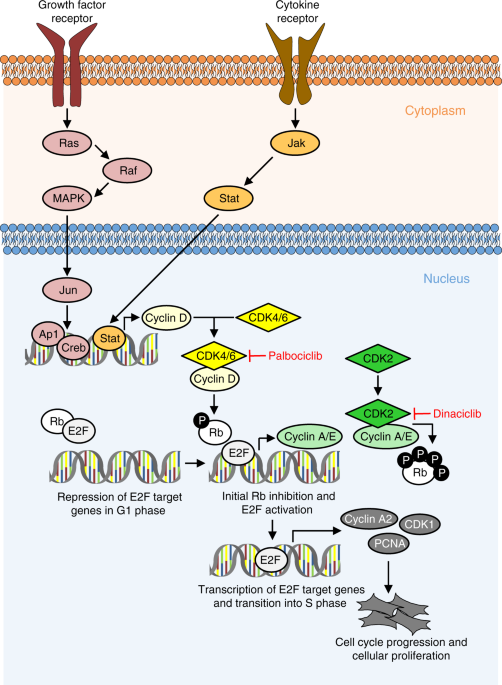 Proposed mechanism of action of palbociclib and dinaciclib in PAH. Multiple growth factors, cytokines, and mitogens induce the activation of cyclin-dependent kinases (CDKs), e.g., by increasing the expression of cyclin D1.Nat Commun.2019May 17;10(1):2204. |