
| Size | Price | Stock | Qty |
|---|---|---|---|
| 2mg |
|
||
| 5mg |
|
||
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| Other Sizes |
|
Purity: =99.46%
SGX-523 (SGX 523; SGX523) is a novel, exquisitely selective, and ATP-competitive inhibitor of Hepatocyte growth factor receptor/Met with potential anticancer activity. With an IC50 of 4 nM, it suppresses the tyrosine kinase Met and shows no effect on BRAFV599E, c-Raf, Abl, or p38α. Numerous cancer types have been linked to MET during their initiation and progression.
| Targets |
c-Met (IC50 = 4 nM)
|
|---|---|
| ln Vitro |
SGX-523 is a member of the c-Met/hepatocyte growth factor receptor (HGFR) tyrosine kinase inhibitor class. Its selectivity may be explained by the way SGX-523 stabilizes MET in a distinct inactive conformation that is unattainable by other protein kinases. While the closely related receptor tyrosine kinase RON is not potently inhibited by SGX523, the purified MET catalytic domain is. With a higher apparent affinity for the less active, unphosphorylated form of MET [MET-KD(0P), with a Ki of 2.7 nM] than the more active phospho-enzyme [MET-KD(3P), with a Ki of 23 nM], SGX523 shows ATP-competitive inhibition. This phenomenon is consistent with preferential binding to an inactive enzyme conformation. At nanomolar concentrations, SGX523 inhibits MET-mediated signaling, cell migration, and proliferation, but even at micromolar concentrations, it has no effect on signaling that is dependent on other protein kinases, including the closely related RON.
|
| ln Vivo |
SGX523 significantly retards the growth of preestablished GTL16 tumors when administered orally at doses of ≥10 mg/kg twice daily. Strongly suppressing the growth of U87MG tumors, SGX523 causes a noticeable regression of U87MG tumors when dosed twice daily at 30 mg/kg. When SGX523 is administered twice a day at a dose of 30 mg/kg, it also lowers tumor MET autophosphorylation levels while slowing the growth of H441 tumors. Tumor xenografts derived from human glioblastoma, lung, and gastric cancers grow less rapidly in response to SGX523's dose-dependent inhibition of MET in vivo, indicating that these tumors are dependent on Catalytic Activity of MET. [1]
|
| Enzyme Assay |
In the presence of 100 mM HEPES (pH 7.5), 0.3 mg/mL poly(Glu-Tyr) peptide substrate, 10 mM MgCl2, 1 mg/mL bovine serum albumin, 5% DMSO, 20 nM MET-KD, and varying concentrations of ATP and SGX523, initial rate constants are measured at 21 °C. Kinase-Glo detection buffer (20 μL) is used to quench the entire reaction volumes (20 μL). A plate-reading luminometer is used to detect luminosity, and nonlinear regression is used to analyze the data.
|
| Cell Assay |
In a 24-well plate, 1 × 103 MDCK cells are seeded per well and incubated for one week at 37 °C with 5% CO2 in MEM and 10% fetal bovine serum. After adding different concentrations of SGX523 and HGF (90 ng/mL), the cells are incubated for an additional 18 hours at 37 °C in a humidified incubator with 5% CO2 before being observed. To study cell migration, A549 cells are plated in 12-well plates (6 × 104 per well) and incubated until confluence. Using a pipette tip, create a channel by scraping the monolayers. The compound is added to the starve medium at different dilutions both with and without HGF (90 ng/mL). After twenty-four hours, the wells are examined for cell migration.
|
| Animal Protocol |
GTL16, U87, or H441 xenografts in Harlan nude mice
60 mg/kg Oral gavage |
| References |
| Molecular Formula |
C18H13N7S
|
|
|---|---|---|
| Molecular Weight |
359.41
|
|
| Exact Mass |
359.10
|
|
| Elemental Analysis |
C, 60.15; H, 3.65; N, 27.28; S, 8.92
|
|
| CAS # |
1022150-57-7
|
|
| Related CAS # |
|
|
| Appearance |
Solid powder
|
|
| SMILES |
CN1C=C(C=N1)C2=NN3C(=NN=C3SC4=CC5=C(C=C4)N=CC=C5)C=C2
|
|
| InChi Key |
BCZUAADEACICHN-UHFFFAOYSA-N
|
|
| InChi Code |
InChI=1S/C18H13N7S/c1-24-11-13(10-20-24)16-6-7-17-21-22-18(25(17)23-16)26-14-4-5-15-12(9-14)3-2-8-19-15/h2-11H,1H3
|
|
| Chemical Name |
6-[[6-(1-methylpyrazol-4-yl)-[1,2,4]triazolo[4,3-b]pyridazin-3-yl]sulfanyl]quinoline
|
|
| Synonyms |
SGX-523; SGX 523; SGX523
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
|
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.7823 mL | 13.9117 mL | 27.8234 mL | |
| 5 mM | 0.5565 mL | 2.7823 mL | 5.5647 mL | |
| 10 mM | 0.2782 mL | 1.3912 mL | 2.7823 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
| NCT Number | Recruitment | interventions | Conditions | Sponsor/Collaborators | Start Date | Phases |
| NCT00606879 | Terminated | Drug: SGX523 Capsules | Advanced Cancer | SGX Pharmaceuticals, Inc. | January 2008 | Phase 1 |
| NCT00607399 | Terminated | Drug: SGX523 Capsules | Advanced Cancer | SGX Pharmaceuticals, Inc. | January 2008 | Phase 1 |
|
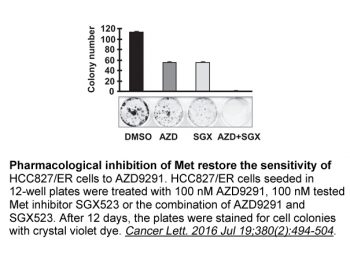 |
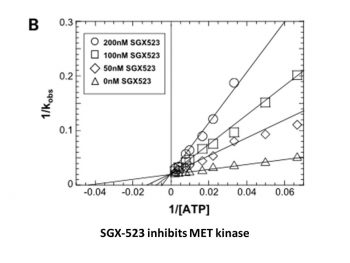 |